Today’s Dietitian
Vol. 18, No. 12, P. 48
Learn About Its Impact on the Immune System, Its Role in Autoimmunity, and Its Importance for Specific Autoimmune Diseases.
Suggested CDR Learning Codes: 2060, 2090, 4040, 5120
Suggested CDR Performance Indicators: 8.1.3; 8.1.5; 8.3.6; 10.4.4
CPE Level: 2
Take this course and earn 2 CEUs on our Continuing Education Learning Library
Under normal circumstances, the immune system protects the body from foreign invaders. However, in some people, the immune system loses tolerance to the body’s own tissues, and immune cells attack and destroy the individual’s cells.1 This self-intolerance and tissue destruction that interrupts normal bodily function is known as autoimmune disease.2
Researchers have identified at least 80 different autoimmune diseases that affect at least 23.5 million Americans.3 The American Autoimmune Related Diseases Association, a nonprofit health agency whose mission is to eradicate autoimmune diseases and alleviate the suffering and the socioeconomic impact, believes both of these numbers may be too low and estimates there may be as many as 140 different diseases with a basis in autoimmunity. The organization also believes there may be as many as 50 million Americans with an autoimmune disease, as the 23.5 million estimate is based on only 24 autoimmune diseases.2 Whichever estimate is used, there appears to be no dispute that the prevalence is increasing.2,3
Although the specific mechanism that causes autoimmune disease is unknown, there are several factors that seem to either trigger or prevent its development. Infections from bacteria or viruses, toxins from microorganisms or the environment, and some medications may trigger an autoimmune response in genetically susceptible individuals.2 Nutrients or drugs that modulate the immune system can help prevent or reverse an autoimmune response.3 Due to its ability to change the immune response, vitamin D is one nutrient that’s believed to positively impact many autoimmune diseases.4
This continuing education course explores the impact of vitamin D on the immune system, explains the role of vitamin D in autoimmunity, and discusses the importance of vitamin D in specific autoimmune diseases based on the latest research.
Brief Introduction to Vitamin D
As presented in the continuing education course “Vitamin D’s Role in Health—Deterministic or Indeterminate?,” vitamin D is a prohormone that humans can either synthesize under certain conditions or consume in food. One form known as vitamin D3 is created in the skin when one is exposed to an adequate amount of ultraviolet B (UVB) radiation.4 Vitamin D3 also can be consumed from foods in which it naturally occurs, including oily fish such as herring, cod liver oil, egg yolks, and sun-exposed mushrooms.5 The other form of vitamin D, known as vitamin D2, is artificially manufactured. It’s used to fortify foods such as milk, orange juice, margarine, and infant formulas.4,5
After being released from the skin or ingested from food, vitamin D travels to the liver, where it’s turned into 25(OH)D, or calcidiol. This hydroxylated version then travels through the circulatory system to numerous tissues, where it’s converted into its active form called calcitriol, or 1alpha,25(OH)2D.6 The cells that produce the enzyme necessary to turn calcidiol into calcitriol include those of the kidneys, muscles, colon, immune system, pancreas, breasts, and brain.5,6
Once calcitriol has been created, it can interact with the vitamin D receptor (VDR) found in numerous cells, which triggers messengers within the cell.6 In addition, vitamin D has been shown to regulate gene transcription.4 Between 200 and 600 genes are primary targets of vitamin D, and as many as 2,100 additional genes are involved in VDR binding.5,6 Given that the VDR is found in cells from the brain and skin to immune cells and gonads, it isn’t surprising that vitamin D has been proven to have an impact on human physiology in numerous ways, including contributing to calcium homeostasis, brain and fetal development, heart function, and innate and adaptive immunity.4,5
Brief Overview of the Immune System
To understand the effect of vitamin D on the immune system, a basic understanding of human immunity is required. As shown in the diagram on page 50, the human immune system is complex, with many factors influencing how the body responds to invading organisms or toxins.1 Immunity to microbes that are on or in the body involves two types of systems: innate immunity and adaptive immunity.7
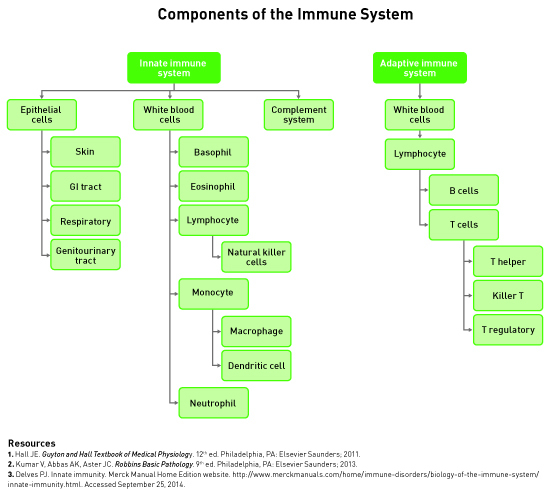
Innate immunity is the natural or native immunity that’s present in a person from birth. The first defense of innate immunity is the physical barrier created by epithelial cells.7 When microbes get past the physical barriers, white blood cells and complement proteins identify and eliminate some of them.8 For example, when tissues indicate that an infection is occurring, a type of white blood cell called a monocyte moves from the blood into the tissue and becomes a macrophage. Macrophages ingest and degrade pathogens using self-produced microbicidal substances.8 Dendritic cells also can consume and destroy pathogens.7 Eosinophils and natural killer cells attach to microbes and release substances into the surrounding area that damage the pathogens.8
In addition to destroying pathogens, the innate immune system triggers other parts of the same system and parts of the adaptive immune system to respond. Dendritic cells, macrophages, and basophils release messenger molecules known as cytokines or inflammatory mediators.8 These chemical messengers travel through the circulatory and lymphatic systems and initiate innate and adaptive immune system activity.7 Innate immune system members, including macrophages, dendritic cells, and complement proteins, carry and present antigens to members of the adaptive immune system. These antigens are pieces of the destroyed microbes or innate immune cells that are presented to members of the adaptive immune system to trigger their involvement.8
The adaptive immune system is so called because it responds to pathogen-associated triggers such as antigens and cytokines. The activated adaptive immune system results in naïve or memory T or B cells maturing and proliferating to eliminate the presented pathogen directly or further activate the innate immune system to destroy the invader. Activated B cells multiply and produce pathogen-specific antibodies, also called immunoglobulins, while T cells differentiate into T helper (Th), T regulatory (Treg), or killer T (KT) cells, depending on the type of stimulation received.1,5 Th cells release cytokines that trigger action by the innate and adaptive immune systems; KT cells directly attack and kill any cell that’s deemed foreign; and Treg cells help to regulate the activity of Th and KT cells and, therefore, the activity of the adaptive immune system.1 It’s worth noting that KT cells also are called cytotoxic T cells and natural KT (NKT) cells, and are different from the natural killer (NK) cells of the innate immune system.
Although this description shouldn’t be considered an exhaustive explanation of the human immune system, it’s sufficient for understanding how vitamin D influences the activity of the innate and adaptive immune systems in the following section.
Vitamin D’s Function in the Immune System
The interactions between vitamin D and the immune system are numerous. First, some immune cells contain both the VDR and the gene CYP27B1, which is responsible for generating the enzyme that activates vitamin D. This means both calcitriol and calcidiol have influential interactions with immune cells because the immune cell can activate and then respond to the activated vitamin D. Differentiated monocytes, B cells, and T cells all express the vitamin D–activating enzyme 25-hydroxyvitamin D-1α-hydroxylase.9 Interestingly, the cytokine interferon γ (IFNγ), released primarily by NK and KT cells, increases the expression of the enzyme in macrophages.4,9 This means that the immune system can self-perpetuate calcitriol-induced activity. It also allows the immune system to achieve the calcitriol level necessary for the immune response without increasing the calcitriol level throughout the entire body.10
In addition to activating vitamin D, almost all immune cells have VDRs, which means vitamin D that has been activated can interact with the VDR on the same or other immune cells in the vicinity.10 Mature and activated T cells, B cells, neutrophils, macrophages, and dendritic cells all express VDRs.10,11 Vitamin D induces the production of cytokines interleukin 4, interleukin 5, and interleukin 10 by dendritic cells, which results in the increased production of Th2 by the adaptive immune system.9 Vitamin D activates macrophages and induces them to increase their production of the antimicrobial peptide cathelicidin, which breaks down pathogens such as Mycobacterium tuberculosis once the macrophage has consumed them.5,6,9 This creates a recursive effect whereby vitamin D both generates more innate immune cells and makes them more effective at destroying harmful organisms.6 On the other hand, vitamin D reduces macrophages’ ability to stimulate the adaptive immune system by preventing the release of proinflammatory mediators and diminishing their ability to present antigens to adaptive immune system cells.9
In addition to increasing the production of Th2 in the adaptive immune system, calcitriol increases the production of Treg cells, which control the adaptive immune system as previously mentioned.9,11 Another way vitamin D controls the immune system is by inhibiting the production of Th1, which means fewer interleukin 2 (IL-2) and IFNγ cytokines are released.9 IL-2 and IFNγ are responsible for further macrophage activation and B cell production of certain antibodies.4,7 Beyond reducing the total numbers of Th1, calcitriol reduces the amount of cytokines produced by Th1 and Th17 cells, diminishing their downstream effect.11 Vitamin D also lessens the involvement of B cells in the immune response by decreasing production of B cells, decreasing their ability to produce antibodies, and triggering apoptosis or programmed cell death.6,9
In a study using mice whose ability to create VDRs was eliminated, Cantorna and colleagues showed that those mice produced more Th1 and Th17 cells than did the control mice, although there was no change in their ability to produce Th2 or Treg cells.11 Also, the VDR knockout mice produced very low numbers of a type 1 or invariant NKT (iNKT) cell, as compared with control mice. Interestingly, in the same study using pregnant mice with VDR-generating capability, if a pregnant mouse was deficient in vitamin D, the offspring didn’t produce as many iNKT cells even after receiving calcitriol supplementation in infancy as did those offspring whose mothers weren’t vitamin D deficient during pregnancy. This suggests that a lack of iNKT cells may be attributable to vitamin D deficiency and/or a lack of VDR within the naïve T cells.
Beyond activity in mature and activated immune cells, some immature immune system cells also have VDRs in their nuclei. Some studies have shown that vitamin D blocks the maturation of dendritic cells.9 Dendritic cells express fewer VDRs as they mature, meaning they’re less responsive to calcitriol once maturation is complete and, therefore, produce fewer proinflammatory cytokines.9,10 Finally, based on the research by Cantorna and colleagues,11 a lack of vitamin D or a deficiency of VDR prevents the maturation of T cells into iNKT cells, thus pushing the development of T cells toward other types, such as Th cells.
Although the cause of the difference is unclear, studies don’t always yield the same results pertaining to calcitriol levels and immune system function. For example, some investigators have shown that calcitriol causes an increase in cytokine production by Th2 cells, while others have shown a decrease.11 While the different effects observed no doubt have to do with the complexity of the immune system and the many factors that influence it, studies generally have shown that vitamin D seems to upregulate the innate immune response and downregulate the adaptive immune response.9 This may mean that vitamin D can prevent excessive inflammation, which is the protective response of the body to harmful invaders, while allowing localized response and resolution primarily by the innate immune system.7,10
Autoimmunity and Specific Autoimmune Diseases
The incidence of autoimmune diseases is increasing throughout the world.9 The association between vitamin D deficiency and autoimmune diseases was first suggested based on the higher prevalence of autoimmune diseases in people living at higher latitudes due to the decreased exposure to sufficient UVB radiation. Epidemiologic studies have confirmed the correlation between the risk of developing an autoimmune disease and vitamin D deficiency.12 For example, higher intake of vitamin D, living at lower altitudes, and increased UVB exposure have been correlated with reduced risk of type 1 diabetes mellitus (T1DM), multiple sclerosis (MS), rheumatoid arthritis (RA), and inflammatory bowel disease (IBD).5 In addition, vitamin D deficiency has been associated with symptom exacerbation in people with an autoimmune disease.9 Because vitamin D has been shown to modulate the immune response, it’s assumed that vitamin D deficiency increases the risk of immune-mediated disease development or symptom exacerbation, although reverse causation can’t be ruled out.13
In one large study using more than 8.6 million hospital records for all of England from 1999 to 2011, Ramagopalan and colleagues showed an association between vitamin D deficiency and diagnosis of autoimmune disease.13 According to the hospital records, 13,260 patients were diagnosed with vitamin D deficiency or associated proxies, such as osteomalacia or rickets, before diagnosis of any autoimmunity. These patients also had significantly more diagnoses of various autoimmune diseases, including Addison’s disease, ankylosing spondylitis, celiac disease, Crohn’s disease, pernicious anemia, primary biliary cirrhosis, and thyrotoxicosis compared with patients who didn’t receive a diagnosis of vitamin D deficiency. The authors acknowledged that reverse causality may explain the vitamin D deficiency in that the autoimmune disease may have been present but not recorded before the admission and before the hypovitaminosis D manifestation. Some autoimmune diseases such as celiac disease may cause malabsorption and lead to vitamin D deficiency, and there may be confounding factors that weren’t available for consideration in the analysis.
Handel and colleagues used an experimental technique for genome sequencing abbreviated as ChIP-seq to determine where the VDR binds with the genome of T cells and whether this binding is affected by levels of serum calcidiol.12 They found that the VDR-binding regions of the T cell genome change based on the level of serum calcidiol. Specifically related to the genes whose sequence variation is associated with autoimmune diseases, study participants with serum calcidiol levels ≥75 nmol/L showed a significant increase in the VDR binding sites on T cell genes compared with study participants whose serum calcidiol levels were below 75 nmol/L. This finding was especially true in genes expressed in the Treg and Th cells. The implication is that higher calcitriol levels may control adaptive immunity-related autoimmune diseases by influencing the genetic activity of T cells.
A review by Waterhouse and colleagues proposed that some autoimmune diseases may be a result of VDR dysfunction caused by intracellular bacteria.14 The proposal is based on the observation that calcidiol levels, the form of vitamin D most often measured, are low in patients with autoimmune diseases such as RA, systemic lupus erythematosus (SLE), and Sjögren’s syndrome, while calcitriol levels are quite high. The treatment involves restoring VDR function with a VDR agonist and bacteriostatic antibiotics, while controlling vitamin D intake and skin synthesis to prevent increasing serum calcitriol. In their review, they found evidence that treatment caused positive responses in subjects with RA, Sjögren’s syndrome, autoimmune thyroid disease (AITD), psoriasis, T1DM, sarcoidosis, and others.
Finally, some studies have found no correlation between serum vitamin D levels and incidence of autoimmune diseases.9 In addition to the possibility of VDR dysfunction described by Waterhouse and colleagues, Antico and colleagues found that polymorphisms in the gene that produces VDRs are associated with increased risk of many autoimmune diseases, including Hashimoto’s disease.9 The relative risk depends on which alleles are present in different classes of genes, with multiple genetic variations conferring various levels of increased risk. Because much of the population is believed to have hypovitaminosis D and genetic variations have been correlated with autoimmune disease, vitamin D deficiency has been suggested as an environmental factor that contributes to, but isn’t primarily responsible for, the onset of autoimmune disease in genetically susceptible individuals.
Due to the varied findings related to vitamin D levels and autoimmune disease incidence, reviewing the literature related to vitamin D and specific autoimmune diseases is necessary to determine the best clinical practice. Following is the latest research regarding the correlation between vitamin D and select autoimmune diseases.
IBD
IBD is a complex disease whose cause is unclear. It’s characterized by chronic inflammation of the gastrointestinal tract and has two forms: Crohn’s disease and ulcerative colitis. IBD is associated with an abnormal immune response that leads to abdominal pain, diarrhea, and weight loss. Researchers have found the inflammatory response in IBD is associated with cytokines from activated macrophages and Th1 cells and T cell proliferation. Vitamin D therapy in IBD patients has been shown to reduce the production of proinflammatory cytokines and modulate seemingly uncontrolled lymphocyte production during IBD inflammation.15
Wang and colleagues researched the association between VDR polymorphisms and IBD risk.16 Their meta-analysis showed that of the four widely evaluated VDR polymorphisms, one (ApaI) is significantly associated with Crohn’s disease risk in all studied populations, one (BsmI) with Crohn’s disease risk in East Asian populations, and one (TaqI) with Crohn’s disease risk in whites. Interestingly, TaqI also is associated with decreased ulcerative colitis risk, especially in whites. Given that the impact of VDR polymorphisms wasn’t consistent across all populations, the reviewers suggest that the VDR genetic variations may interact with other genetic variations or environmental factors that result in IBD.
In another review, Garg and colleagues noted that many patients with IBD may be vitamin D deficient due to malabsorption that occurs in gastrointestinal epithelial diseases or to reduced sunlight exposure and physical activity, especially during times of acute inflammation.17 However, they also found that epidemiologic studies illustrate that ulcerative colitis onset and Crohn’s disease exacerbations are greater in winter; incidence and prevalence of IBD increase at higher latitudes; and there’s a lower Crohn’s disease risk with higher predicted serum vitamin D.
These correlations suggest that maintaining an adequate serum vitamin D level may reduce the risk and flare-ups of IBD.
MS
MS is an inflammatory disorder in which T cells progressively target the central nervous system.11,18 MS relapse is more common in the winter; babies born during the winter are more likely to develop MS later perhaps due to maternal vitamin D deficiency; and lower vitamin D levels are associated with increased disability and clinical activity, especially in patients with increased clinical activity, disability, and risk of relapse.9
An animal model of MS called experimental autoimmune encephalomyelitis (EAE) revealed that Th1 and Th17 cells attack the animals’ central nervous system. Under normal circumstances, the iNKT and Treg cells would regulate the activity of Th1 and Th17 cells. However, patients with MS have been shown to have fewer iNKT and Treg cells. In fact, symptom remission is correlated with increased production and activity of these cells.11 As mentioned previously, calcitriol upregulates the production of iNKT and Treg cells.9,11
Farias and colleagues showed that EAE rats treated with 10 or 15 μg/kg/day of vitamin D3 showed a significant increase in the production of Treg cells and a significant decrease in the production of autoreactive T cells, which are T cells that attack human cells as a result of tissue-derived antigens.19 It’s worth noting that although the number of cells that infiltrated the central nervous system decreased, the number of inflammatory sites within the central nervous system wasn’t significantly different in the vitamin D3-treated rats.
Despite the seemingly positive epidemiologic and animal studies, a study by Kampman and colleagues showed no beneficial effects on clinical outcomes, including disability, for 35 patients suffering from MS with 96 weekly supplementations of 20,000 IU vitamin D3 when compared with 33 controls.20 In addition, James and colleagues conducted a meta-analysis that showed no significant association between MS relapses and high-dose or long-term vitamin D treatment. The researchers noted that there were methodological limitations to the studies, such as short duration or small sample size.18 It’s also possible that VDR variations in MS patients, which were noted but not taken into consideration in all studies, could explain the variable response to vitamin D supplementation.18
Given the seemingly conflicting results, further research is required to determine when vitamin D therapy is useful for patients suffering from MS.
Psoriasis
Psoriasis is an inflammatory disorder of the skin in which keratinocytes proliferate and differentiate at a higher-than-normal rate, leading to lesions, known as plaque, that cover the inflamed skin. The pathogenesis includes a dysregulated interaction between epidermal skin cells and T cells that results in the recruitment of additional proinflammatory immune cells.21 In a cross-sectional study, Gisondi and colleagues found that patients with chronic plaque psoriasis have significantly lower levels of serum vitamin D than do healthy controls.22 Also, regression showed that the hypovitaminosis D of psoriasis patients is independent of age, gender, BMI, serum calcium levels, serum parathyroid hormone levels, and season. The authors noted that a causal relationship can’t be determined from the study.
For many years, narrowband UVB (NB-UVB) radiation has been used to treat psoriasis. Based on the fact that exposure to UVB radiation from sunlight causes the production of vitamin D, it isn’t surprising that studies of NB-UVB–treated psoriasis patients have shown a statistically significant increase in calcidiol following consistent and frequent NB-UVB treatment. NB-UVB treatment and increased calcidiol levels are associated with clinical improvement of the disease.23
In patients for whom NB-UVB treatment is inconvenient, the topical vitamin D analogue calcipotriol has been shown to reduce keratinocyte proliferation. Interestingly, treatments that increase serum calcitriol also increase the production of cathelicidin by macrophages, which is part of the innate immune response.23 Because some successful treatments increase the potency of innate immune cells, psoriasis activity doesn’t appear to be induced by the innate immune system at all.
It’s worth noting that a meta-analysis performed by Stefanic and colleagues showed no association between the widely studied VDR polymorphisms and psoriasis.24 In addition, recent studies regarding the impact of oral vitamin D intake on psoriasis are lacking. That said, studies continue to show that adequate serum vitamin D appears to prevent and reduce the duration of psoriatic flare-ups.24
RA
RA is a chronic autoimmune disease that primarily affects synovial joints, which are a type of moveable joint in the wrists, elbows, shoulders, hips, and knees. Flare-ups are characterized by the synovial infiltration of innate and adaptive immune cells, which ultimately leads to tissue damage.25 In their meta-analysis, Song and colleagues found that total vitamin D intake from food and supplements is significantly inversely associated with RA incidence.25 In fact, study participants with the highest vitamin D intake had a 24.2% reduced risk of RA development compared with those who consumed the lowest amount.
Turhanoglu and colleagues found no statistically significant difference in serum vitamin D levels between healthy controls and RA patients, as the average calcidiol level showed hypovitaminosis D in both groups.26 However, within the RA patient group, they found those with the least disease activity had the highest vitamin D levels, while people with the most disease activity as measured on three different scales had the lowest vitamin D levels.
Yarwood and colleagues discovered that 37% of genetic RA regions contain VDRs, which is a higher percentage of VDR binding sites than the rest of the genome.27 This lends further support to the observation that vitamin D levels impact RA activity, but likely only in those with certain genetic sequencing.
Given that most studies show an inverse association between serum vitamin D and RA activity,26,27 patients with RA or who are genetically susceptible to RA may benefit from sufficient serum vitamin D levels obtained via food, supplements, and/or UVB radiation.
SLE
SLE has been defined as an autoimmune disease in which the immune system attacks various healthy tissues, including the skin, joints, kidneys, and brain. It’s more common in women than men; it also affects African Americans and Asians more than other populations. The primary symptoms include pain and swelling of the joints in the hands, wrists, and knees.28 Studies have been somewhat inconclusive about the role of vitamin D in SLE activity. Some have shown an inverse relationship between vitamin D levels and SLE activity, while others have shown no association.29
Bonakdar and colleagues found that most women (75%) with newly diagnosed SLE were deficient in vitamin D.30 Their disease activity, as measured by the British Isles Lupus Assessment Group index, was inversely related to the level of vitamin D, even when controlling the analysis for age, BMI, sun exposure, photosensitivity, sunscreen use, and physical activity. Of the various factors measured by the index, the presence of SLE-related antibodies and complement proteins were two of the most significant factors correlated with vitamin D deficiency.
Casella and colleagues also showed an inverse correlation between serum vitamin D levels and disease activity.29 In addition, they found that patients with juvenile onset SLE may be vitamin D-deficient even when receiving vitamin D supplements. They suggest that glucocorticoid treatment may affect the metabolism of vitamin D, resulting in hypovitaminosis D despite supplementation (average dose 429 ± 410 IU). In fact, there was no significant difference in serum calcidiol levels between juvenile onset SLE patients who received vitamin D supplements and those who didn’t.
Some animal models have shown that vitamin D supplementation has either a positive or a negative effect on disease activity, depending on serum calcium levels. It appears that serum calcium levels must be sufficient for vitamin D supplementation to positively influence disease activity.29 These results remain to be proven in humans.
Given the available information, patients with SLE may benefit from maintaining sufficient levels of serum calcidiol. Achieving an adequate level may require supplementation above standard amounts, depending on whether the treatment being received affects vitamin D metabolism or for those who exhibit sensitivity to sunlight and therefore can’t get it from UVB exposure. In addition, based on the animal models, it may be worth monitoring patients’ serum calcium levels in conjunction with calcidiol levels.
AITD
AITD occurs when the immune system attacks the tissue of the thyroid leading to either hyperthyroidism, called Graves’ disease, or hypothyroidism, called Hashimoto’s disease.31 In a meta-analysis, Feng and colleagues found that the BsmI and TaqI VDR polymorphisms are significantly associated with AITD risk.31 The other two common VDR polymorphisms, ApaI and FokI, weren’t associated with AITD risk. In addition, other studies have shown that polymorphisms in the vitamin D binding (VDB) protein that carries calcidiol through the blood also are associated with AITD.32
Effraimidis and colleagues studied 521 women who showed no signs of thyroid antibodies, didn’t have hypo- or hyperthyroidism, and had a first- or second-degree relative with documented AITD.33 During the follow-up period of up to five years, 67 women developed thyroid antibodies. Serum calcidiol was <50 nmol/L for more of the 67 women at baseline (49%) than at the time thyroid antibodies were found (52%). This was also true for the matched controls (34% vs 55%). This implies that vitamin D deficiency does not cause AITD onset, perhaps either at all or without other contributing factors.
Yasuda and colleagues showed that patients with active Graves’ disease had lower calcidiol levels than did either Graves’ disease patients in remission or the control group.32 However, they didn’t find a significant correlation between serum calcidiol and serum thyroid antibody levels. On the other hand, Bozkurt and colleagues studied patients with Hashimoto’s disease and appropriate levels of thyroid hormone, either alone or with thyroid treatment.34 They found that the longer the patient had the disease, the smaller the thyroid, and the more thyroid antibodies in the blood, the lower the calcidiol level. Moreover, patients who were taking thyroid replacement hormone had lower calcidiol levels, which may be attributable to the chronic state of Hashimoto’s disease as indicated by the small size of the thyroid and number of thyroid antibodies.
Given the available information, it seems that vitamin D level alone may not be responsible for the onset or activity of Graves’ disease, but may be related to Hashimoto’s disease. However, polymorphisms in the VDR or VDB may be related to disease incidence for both classes of AITD.
Unfortunately, the efficacy of vitamin D supplementation to prevent Hashimoto’s disease has been shown only in animal models.35 Furthermore, no research could be found regarding vitamin D supplementation to reverse AITD.
T1DM
T1DM is an autoimmune disease in which immune cells attack the pancreatic beta cells that are responsible for producing insulin. Most often the disease is diagnosed in patients before age 30, with the highest incidence around puberty.36
Epidemiologic studies and meta-analyses suggest that there appears to be a reduction in T1DM risk following regular vitamin D supplementation in early life.37 For example, infants in one study who were given at least 2,000 IU/day had 88% less risk of developing T1DM by age 32, while in another study, vitamin D supplementation early in life was associated with a 33% reduction of T1DM risk in children.37,38 More specifically, some studies have shown that supplementation between the ages of 7 and 12 months reduces the risk of developing T1DM more than supplementation between the ages of 0 and 6 months. This is likely due to the increased development that occurs in the adaptive immune system during the 7- to 12-month period of life.9 It’s also worth noting that there appears to be a dose-dependent response, with higher doses of vitamin D supplementation resulting in lower risk of T1DM development.9
Azab and colleagues found that the number of T1DM patients who were deficient in vitamin D was significantly greater when compared with controls (55% vs 30%).36 In addition, significantly more T1DM patients had secondary hyperparathyroidism (a parathyroid gland response to hypocalcemia), than those without T1DM. It’s worth noting that vitamin D levels within the T1DM group were negatively correlated with BMI and positively correlated with disease duration. That said, the amount of vitamin D needed to affect the disease is unknown. In one study, low doses of calcitriol showed little to no improvement in insulin needs for T1DM patients.37
Given the available information, the best approach for reducing risk of T1DM as it relates to vitamin D is likely to ensure sufficient supplementation in early life and maintain appropriate vitamin D levels, should T1DM occur.
Conclusion
Vitamin D is a prohormone humans synthesize in their skin or obtain through foods or supplements containing vitamin D.4 The VDB protein carries the inactive version through the circulatory system so it can complete a two-step activation process involving the liver and the cells of the kidneys, immune system, and other tissues. The active form, called calcitriol, interacts with the VDR on many cells to elicit a hormonal or genetic response.6
Many cells of the adaptive and innate immune systems have VDRs.10 Under most circumstances, vitamin D has been shown to upregulate the activity of the innate immune system and downregulate the activity of the adaptive immune system.9 By regulating the immune system in this way, vitamin D appears to prevent excessive inflammation while allowing a localized immune response.7,10
Hypovitaminosis D has been suggested as a contributing factor to autoimmune disease as the prevalence of autoimmunity is greater at higher latitudes.12 Many studies have shown a correlation between autoimmune disease and vitamin D levels.5,13 In addition, research has shown that specific genetic variations associated with autoimmune disease have interaction with VDRs.14 Unfortunately, causality is difficult to prove, and for some autoimmune diseases, such as those of the GI tract, reverse causality can’t be eliminated.13 However, vitamin D supplementation generally has been shown to be beneficial in reducing autoimmune disease severity, even though the data are insufficient thus far to establish a causal effect of vitamin D deficiency and autoimmune disease incidence.9
Regarding the selected autoimmune diseases covered here, research seems to support the link between vitamin D deficiency and the onset and/or exacerbation of IBD, psoriasis, RA, SLE, and Hashimoto’s disease. Sufficient vitamin D supplementation in infancy appears to significantly reduce the risk of T1DM, while serum vitamin D levels aren’t conclusively linked with either MS or Graves’ disease. It’s important to remember that ethnic origin, time of year, and disability status have a significant impact on serum vitamin D levels, and these should be considered as confounding factors in a person diagnosed with an autoimmune disease.9 Given the current evidence, dietitians are encouraged to ensure vitamin D intake is sufficient to maintain adequate serum calcidiol levels in clients who have been diagnosed with or have a family history of many autoimmune diseases.
As presented in the continuing education course “Vitamin D’s Role in Health—Deterministic or Indeterminate?,” some individuals may need a dietary intake of as much as 1,500 IU of vitamin D per day to ensure adequate maintenance of serum calcidiol levels. The recommended intake depends on an individual’s exposure to effective sunlight and the person’s ability to absorb, metabolize, and utilize dietary sources. Dietary sources of vitamin D are limited and include cod liver oil, maitake mushrooms, swordfish, salmon, and fortified dairy products. If sun exposure and dietary sources are insufficient, supplements may be consumed as straight vitamin D or in combination with other vitamins and minerals in multivitamins.
— Stephanie Dunne, MS, RD, is the founder of Nutrition. QED., a private practice in New York City, and a freelance writer and speaker.
Learning Objectives
After completing this continuing education course, nutrition professionals should be better able to:
1. Define the basics of innate and adaptive immunity.
2. Evaluate the involvement of vitamin D with the immune system.
3. Translate the impact of vitamin D on specific autoimmune diseases.
CPE Monthly Examination
1. In which organ does the first step of vitamin D activation occur?
a. Spleen
b. Kidneys
c. Liver
d. Brain
2. Which of the following immune cells produces cathelicidin?
a. Macrophages
b. Killer T cells
c. Dendritic cells
d. Basophils
3. Immunity is achieved by the response of which two systems?
a. White blood cells and complement proteins
b. Innate and adaptive immunity
c. Natural and responsive immunity
d. Adjustable and native immunity
4. Which of the following differentiated T cells modulate the immune system?
a. Th2
b. Killer T
c. Th17
d. T regulatory
5. In which season are onset and flare-ups of inflammatory bowel disease most common?
a. Spring
b. Summer
c. Fall
d. Winter
6. Rheumatoid arthritis affects which type of joints?
a. Synovial
b. Cartilagenous
c. Fibrous
d. Condyloid
7. Which of the following vitamin D analogues has been successfully used to treat psoriasis?
a. Calcitriol
b. Dihydrotachysterol2
c. 22‐oxacalcitriol
d. Calcipotriol
8. Which of the following immune cells are upregulated by calcitriol, but downregulated by multiple sclerosis?
a. Th1
b. Th2
c. Natural killer
d. Invariant natural killer T
9. Hyperthyroidism associated with autoimmune thyroid disease is referred to as which disease?
a. Thyroiditis
b. Hashimoto’s disease
c. Graves’ disease
d. Goiter
10. Vitamin D supplementation during which stage of life best reduces risk of type 1 diabetes mellitus?
a. 0 to 6 months
b. 7 to 12 months
c. 3 to 5 years
d. Puberty
References
1. Hall JE. Guyton and Hall Textbook of Medical Physiology. 12th ed. Philadelphia, PA: Elsevier Saunders; 2011.
2. Questions and answers. American Autoimmune Related Diseases Association website. http://www.aarda.org/autoimmune-information/questions-and-answers/. Accessed December 3, 2014.
3. Autoimmune diseases fact sheet. Office on Women’s Health website. http://womenshealth.gov/publications/our-publications/fact-sheet/autoimmune-diseases.html. Updated July 16, 2012. Accessed December 3, 2014.
4. Jovicic S, Ignjatovic S, Majikic-Singh N. Biochemistry and metabolism of vitamin D. J Med Biochem. 2012;31(4):309-315.
5. Holick MF. The d-lightful vitamin D for health. J Med Biochem. 2013;32(1):1-10.
6. Battault S, Whiting SJ, Peltier SL, Sadrin S, Gerber G, Maixent JM. Vitamin D metabolism, functions and needs: from science to health claims. Eur J Nutr. 2013;52(2):429-441.
7. Kumar V, Abbas AK, Aster JC. Robbins Basic Pathology. 9th ed. Philadelphia, PA: Elsevier Saunders; 2013.
8. Delves PJ. Innate immunity. Merck Manual Home Edition website. http://www.merckmanuals.com/home/immune-disorders/biology-of-the-immune-system/innate-immunity.html. Accessed September 25, 2014.
9. Antico A, Tampoia M, Tozzoli R, Bizzaro N. Can supplementation with vitamin D reduce the risk or modify the course of autoimmune diseases? A systematic review of the literature. Autoimmun Rev. 2012;12(2):127-136.
10. Baeke F, Gysemans C, Korf H, Mathieu C. Vitamin D insufficiency: implications for the immune system. Pediatr Nephrol. 2010;25(9):1597-1606.
11. Cantorna MT, Zhao J, Yang L. Symposium 3: vitamin D and immune function: from pregnancy to adolescence: vitamin D, invariant natural killer T-cells and experimental autoimmune disease. Proc Nutr Soc. 2012;71(1):62-66.
12. Handel AE, Sandve GK, Disanto G, et al. Vitamin D receptor ChIP-seq in primary CD4+ cells: relationship to serum 25-hydroxyvitamin D levels and autoimmune disease. BMC Med. 2013;11(1):163.
13. Ramagopalan SV, Goldacre R, Disanto G, Giovannoni G, Goldacre MJ. Hospital admissions for vitamin D related conditions and subsequent immune-mediated disease: record-linkage studies. BMC Med. 2013;11:171.
14. Waterhouse JC, Perez TH, Albert PJ. Reversing bacteria-induced vitamin D receptor dysfunction is key to autoimmune disease. Ann N Y Acad Sci. 2009;1173:757-765.
15. Reich KM, Fedorak RN, Madsen K, Kroeker KI. Vitamin D improves inflammatory bowel disease outcomes: basic science and clinical review. World J Gastroenterol. 2014;20(17):4934-4947.
16. Wang L, Wang ZT, Hu JJ, Fan R, Zhou J, Zhong J. Polymorphisms of the vitamin D receptor gene and the risk of inflammatory bowel disease: a meta-analysis. Genet Mol Res. 2014;13(2):2598-2610.
17. Garg M, Lubel JS, Sparrow MP, Holt SG, Gibson PR. Review article: vitamin D and inflammatory bowel disease — established concepts and future directions. Aliment Pharmacol Ther. 2012;36(4):324-344.
18. James E, Dobson R, Kuhle J, Baker D, Giovannoni G, Ramagopalan SV. The effect of vitamin D-related interventions on multiple sclerosis relapses: a meta-analysis. Mult Scler. 2013;19(12):1571-1579.
19. Farias AS, Spagnol GS, Bordeaux-Rego P, et al. Vitamin D3 induces IDO+ tolerogenic DCs and enhances Treg, reducing the severity of EAE. CNS Neurosci Ther. 2013;19(4):269-277.
20. Kampman MT, Steffensen LH, Mellgren SI, Jorgensen L. Effect of vitamin D3 supplementation on relapses, disease progression, and measures of function in persons with multiple sclerosis: exploratory outcomes from a double-blind randomised controlled trial. Mult Scler. 2012;18(8):1144-1151.
21. Hegyi Z, Zwicker S, Bureik D, et al. Vitamin D analog calcipotriol suppresses the Th17 cytokine-induced proinflammatory S100 “arlamins” psoriasin (S100A7) and koebnerisin (S100A15) in psoriasis. J Invest Dermatol. 2012;132(5):1416-1424.
22. Gisondi P, Rossini M, Di Cesare A, et al. Vitamin D status in patients with chronic plaque psoriasis. Br J Dermatol. 2012;166(3):505-510.
23. Al-Mutairi N, Shaaban D. Effect of narrowband ultraviolet B therapy on serum vitamin D and cathelicidin (LL-37) in patients with chronic plaque psoriasis. J Cutan Med Surg. 2014;18(1):43-48.
24. Stefanic M, Rucevic I, Barisic-Drusko V. Meta-analysis of vitamin D receptor polymorphisms and psoriasis risk. Int J Dermatol. 2013;52(6):705-710.
25. Song GG, Bae SC, Lee YH. Association between vitamin D intake and the risk of rheumatoid arthritis: a meta-analysis. Clin Rheumatol. 2012;31(12):1733-1739.
26. Turhanoğlu AD, Güler H, Yönden Z, Aslan F, Mansuroglu A, Ozer C. The relationship between vitamin D and disease activity and functional health status in rheumatoid arthritis. Rheumatol Int. 2011;31(7):911-914.
27. Yarwood A, Martin P, Bowes J, et al. Enrichment of vitamin D response elements in RA-associated loci supports a role for vitamin D in the pathogenesis of RA. Genes Immun. 2013;14(5):325-329.
28. Systemic lupus erythematosus. MedlinePlus website. http://www.nlm.nih.gov/medlineplus/ency/article/000435.htm. Updated January 20, 2015. Accessed November 9, 2015.
29. Casella CB, Seguro LP, Takayama L, Medeiros D, Bonfa E, Pereira RM. Juvenile onset systemic lupus erythematosus: a possible role for vitamin D in disease status and bone health. Lupus. 2012;21(12):1335-1342.
30. Bonakdar ZS, Jahanshahifar L, Jahanshahifar F, Gholamrezai A. Vitamin D deficiency and its association with disease activity in new cases of systemic lupus erythematosus. Lupus. 2011;20(11):1155-1160.
31. Feng M, Li H, Chen SF, Li WF, Zhang FB. Polymorphisms in the vitamin D receptor gene and risk of autoimmune thyroid diseases: a meta-analysis. Endocrine. 2013;43(2):318-326.
32. Yasuda T, Okamoto Y, Hamada N, et al. Serum vitamin D levels are decreased in patients without remission of Graves’ disease. Endocrine. 2013;43(1):230-232.
33. Effraimidis G, Badenhoop K, Tijssen JG, Wiersinga WM. Vitamin D deficiency is not associated with early stages of thyroid autoimmunity. Eur J Endocrinol. 2012;167(1):43-48.
34. Bozkurt NC, Karbek B, Ucan B, et al. The association between severity of vitamin D deficiency and Hashimoto’s thyroiditis. Endocr Pract. 2013;19(3):479-484.
35. Mazokopakis EE, Kotsiris DA. Hashimoto’s autoimmune thyroiditis and vitamin D deficiency. Current aspects. Hell J Nucl Med. 2014;17(1):37-40.
36. Azab SF, Saleh SH, Elsaeed WF, Abdelsalam SM, Ali AA, Esh AM. Vitamin D status in diabetic Egyptian children and adolescents: a case-control study. Ital J Pediatr. 2013;39:73.
37. Takiishi T, Van Belle T, Gysemans C, Mathieu C. Effects of vitamin D on antigen-specific and non-antigen-specific immune modulation: relevance for type 1 diabetes. Pediatr Diabetes. 2013;14(2):81-89.
38. Holick MF, Binkley NC, Bischoff-Ferrari HA, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911-1930.


